Flame spectra
Flame spectra

Overview:
Flame spectra can give a lot of information about the quantum effects occuring in the reaction zones
What is flame spectra?
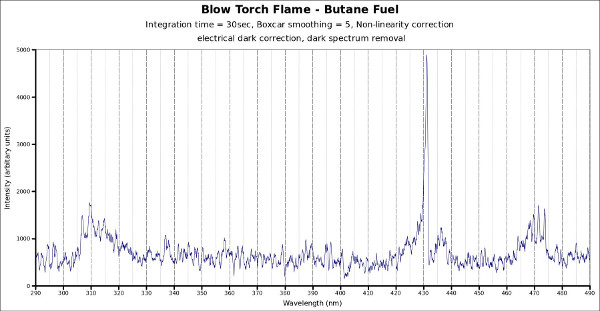
The chart below shows the spectra recorded for a butane blow torch flame. The flame is shown in the photograph below. The flame was at atmospheric pressure.

It can be seen that two peaks exist one at approximately 308nm and the other at approximately 430nm. These peaks correspond to the emissions of two key radicals within the flame front. They are the OH and CH radicals. These radicals are short lived molecules that are formed in the complex chemical breakdown and reformations within the flame front. The reason these are important is that they are highly energetic and try to re-combine with other molecules to try to reduce their energy state. In so doing they radiate visible light energy. Because of quantum mechanical effects there are only discrete wavelengths over which the light may be emitted.
It is possible to infer a great deal of information on the chemical reaction processes inside the combustor provided the light can be collected and a detector of high enough sensitivity is used. However, the sensitivity needed is pushing the limits of currently available spectrometers. The chart below was taken using a spectrometer configured for the highest possible sensitivity. In this case, the signal to noise ratio was not very favourable.
Recently, it has been established that the ratio between the intensities of the 308 and 430nm emissions correlate to flame air to fuel ratios. This is the basis for the spectrometer probe shown in the examples page. The system is provided to clients as a turn-key system, or on-site measurements can be done for the client.